From lab to jab: The COVID-19 vaccine journey
Vaccines are considered one of the greatest developments in modern science.
08 April 2021

From lab to jab: The COVID-19 vaccine journey
Vaccines are considered one of the greatest developments in modern science.
08 April 2021
Over the years the world has seen vaccines save millions of lives, and prevent countless infections from diseases that used to plague society such as polio, smallpox, measles and Spanish flu.
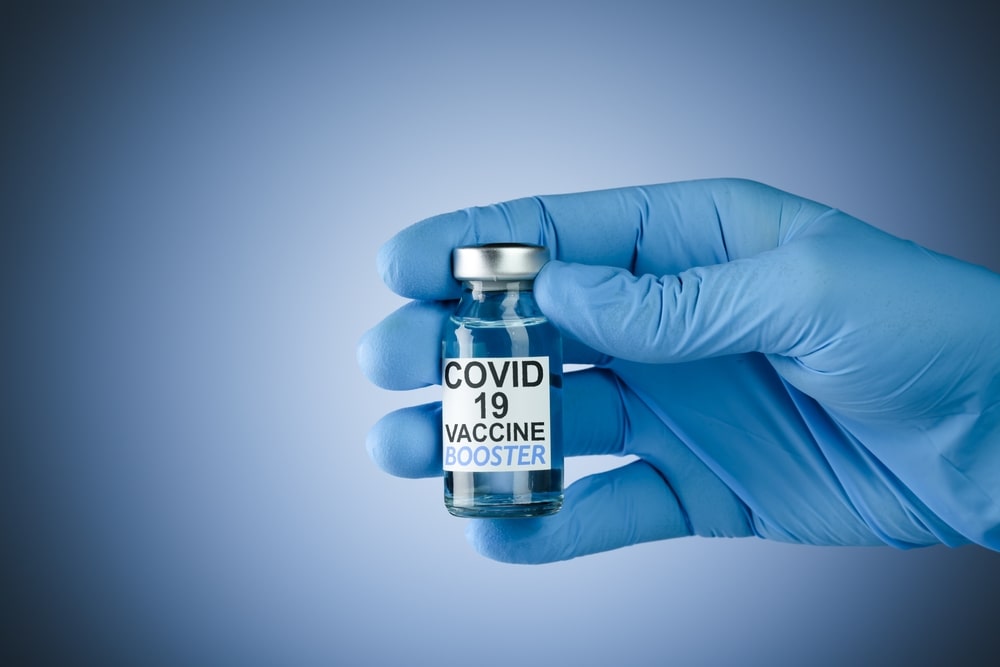
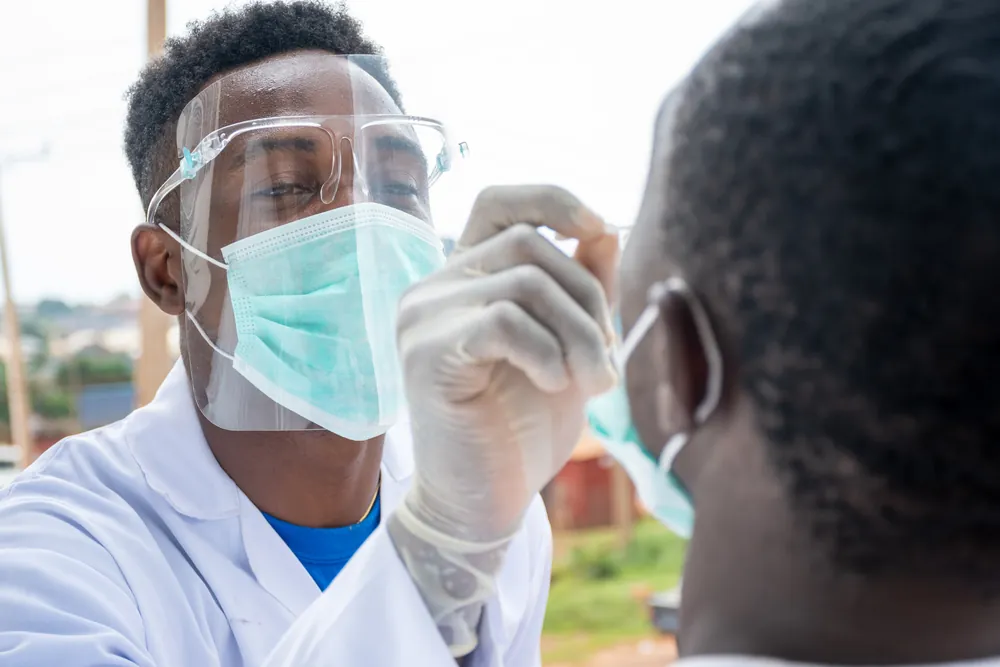
Last month, South Africa received its first batch of Johnson & Johnson COVID-19 vaccines, and began vaccinating frontline healthcare workers immediately. However, while South African healthcare workers line up eagerly to get their vaccines, experts are concerned that many ordinary South Africans are sceptical about the vaccine, especially given how quickly it was developed.
Understanding the journey that our COVID-19 vaccines have taken – from initial research to rollout across the globe – may help South Africans feel more secure about the vaccines that will soon be available to them.
Here are nine key milestones the COVID-19 vaccines will have passed before they make their way into our bodies:
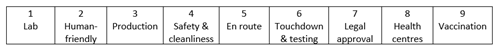
1. Born in the lab:
COVID-19 vaccines, like all other vaccines before it, began life in a laboratory. Thanks to technology advancements, scientists from all over the world collaborated in their research, bringing together the world’s best minds to develop our defence against COVID-19. A key part of the scientific research process was determining which genetic variant of COVID-19 different geographical regions were dealing with. Scientists combined their understanding of the different COVID-19 variants, then tested the vaccine carefully for efficacy.
2. Human friendly:
The vaccine was tested – safely – on humans across continents. Once the vaccine had been developed in the lab, scientists began to test how effective the vaccines are on humans. Thousands of volunteers from around the world signed up to help test the vaccine, and ensure it is safe and works properly for others. The results of these trials were sent to international medical approval bodies to be 100% sure they could be trusted. Only vaccines that passed this critical step moved into production.
3. Beginning production:
The COVID-19 vaccines took around three months to produce on a large scale, and during this process, the vaccines were once again quality checked.
4. Keeping the vaccines clean and safe:
Getting the COVID-19 vaccines around the world is a messy process – and the vaccines themselves need to be kept safe and clean. Once produced at factories, the vaccine liquid is placed into specialised, clean bottles at the point of production and placed into cold storage.
5. En route to South Africa:
At this point, once the vaccines are fully packaged, they are ready to begin their journey to South Africa. However, the vaccines have to be kept cold throughout their travels. The cold chain is thus absolutely critical to ensuring the vaccines are still effective by the time they reach patients.
6. Touchdown – and more testing:
Once COVID-19 vaccines have been received in South Africa, they are transferred into a cold storage truck that will take them to the National Control Laboratory (NCL) in Bloemfontein, where they will receive further testing by some of SA’s leading scientists. The NCL is the only vaccine testing laboratory in the country accredited to do vaccine testing for the World Health Organisation.
7. Legal approval:
Once the NCL has tested the vaccines and has confirmed they are safe for injection, the SA Health Products Regulatory Authority (SAHPRA) provides final, legal approval for the vaccines, meaning that they can be administered to people in South Africa.
All medication in South Africa must receive approval from SAHPRA before it can be given to patients.
8. Arrival at healthcare facilities:
The legally approved vaccines are then transported in refrigerated vehicles to hospitals, pharmacies, and other vaccination sites across the country, where trained staff ensure they are stored correctly (in cold storage) and are ready to be given to patients.
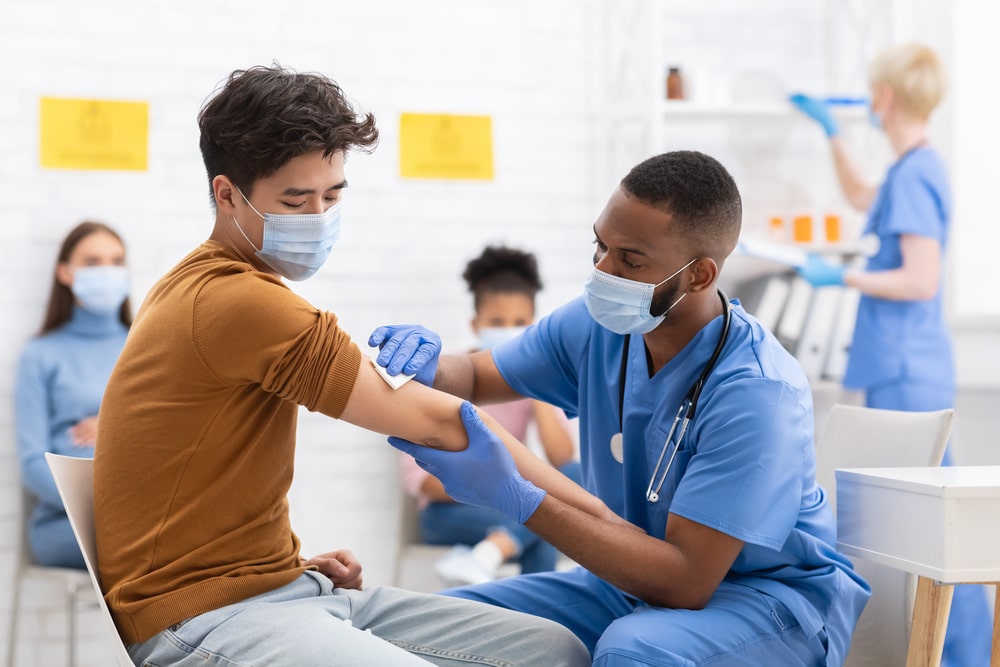
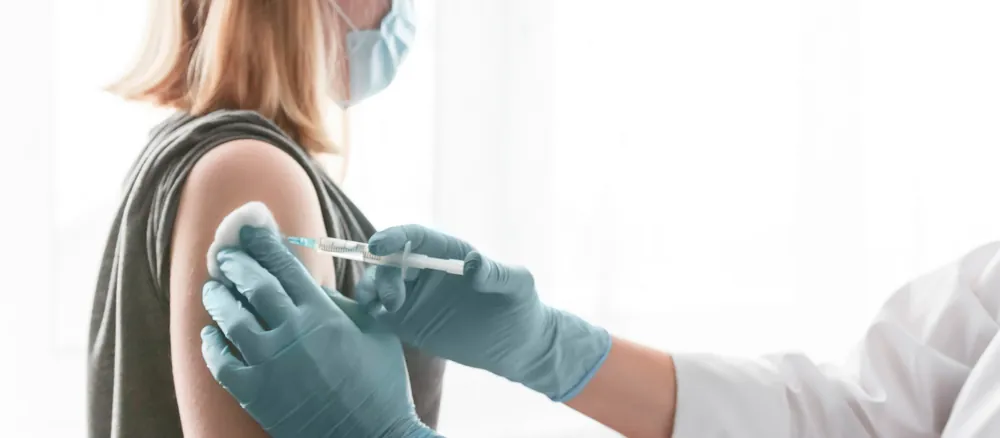
9. Vaccination!
Approved vaccines are administered to patients by an injection into the patient’s upper arm, providing additional protection against the coronavirus to each person who is vaccinated.
Look out for further updates around the COVID-19 vaccination process.